ADU��U3O8�ܽ�Һ��ȡ���ܶԱ�
������������Ұ�������ģ�κ ��
���к���0������˾��һ�ֹ�˾������ ���� 732850��
ժ Ҫ����30%TBP-����ú��Ϊ��ȡ���������˸�Ũ���˵��������(ADU)��U3O8�����ܽ�Һ����ȡЧ����ʵ����������ADU�ܽ�Һ����ȡЧ������U3O8�ܽ�Һ��2�������ܽ�Һ�������ʾ�����Ũ�ȵ����������ߺ����½�����Ũ��Ϊ430 g/Lʱ������������95.43%��90.34%���������ʾ�����ȵ����Ӷ����ӣ�����ͬ�����ADU�������ʽ�U3O8��Լ5%�������Ϊ0~2.5 mol/L��U3O8�ܽ�Һ������������ADU�졣7����ȡ��2���ܽ�Һ����ˮ���е���Ũ�ȷֱ�Ϊ4.42 mg/L��43.42 mg/L��2���ܽ�Һ���е�����ȡ��Ȼ�����ȡҺ���з��ͣ�����Һ�����ʺ����������������ADU�ܽ�Һ������ȥ���ʣ�91%������U3O8�ܽ�Һ������ȥ���ʣ��������ADU����ȡЧ���Ϻá�
�ؼ��ʣ�������������ȡ��������泥�U3O8
����飺��������1989�������У����������ˣ�������ʦ��˶ʿ���˻���רҵ
Extraction performance of nitric solution of ADU and U3O8
Hou Yanlong, Wei Gang, Ma Xiaowen
��The First Filiale of 404 Company Limited, CNNC, Lanzhou 732850, China��
Abstract: The extraction performance of the high concentration uranium solution, including the nitric solution of (NH4)2U2O7 and U3O8, was investigated by 30%TBP-kerosene as organic phase. The results show that the extraction effect of ADU nitric acid solution is better than that of U3O8 solution. The uranium yield of two nitric acid solutions increased first and then decreased slowly with the increase of uranium concentration. The recovery ratio of uranium is 95.43% ((NH4)2U2O7) and 90.34% (U3O8) under 430 g U/L. For the two solutions, the recovery ratio of uranium increases as the flow ratio increases. Moreover, the recovery ratio of (NH4)2U2O7 nitric solution is about 5% higher than that of U3O8 nitric solution. In the experimental acidity range of 0-2.5 mol/L, the uranium yield of two solutions increases with the increase of acidity. And the increase of U3O8 nitric solution is faster than that of (NH4)2U2O7 nitric solution. After 7th extraction, the uranium concentration of raffinate for (NH4)2U2O7 nitric solution and U3O8 nitric solution is 4.42 mg/L and 43.42 mg/L. Meanwhile, two nitric solutions were extracted by single-stage extraction and then stripped. The removal rate of ADU impurities is over 91% by analyzing the content of impurities in the stripping solution. The impurity removing efficiency of ADU is higher than U3O8 solution. The performance of ADU is better than U3O8.
Key words: uranyl nitrate; extraction; ammonium diuranate; triuranium octaoxide
Ŀǰ�����ڹ��ڶ���Դ���������ӣ��˵���Ϊһ������Ч����Դ�������������ǵ����ӣ����ģҲ�ڲ�������[1-2]���ں��ܺ�ƽ�����У�ˮұ����˴�����Ϊ��ȼ��ѭ������ȱ�ٵĹ���[3]��������Դ�������õ���Ҫ����[4]��
�˿�ʯ��ˮұ��������ĥ����ȡ��������һϵ�й��գ����յõ���Ʒ�˿�Ũ������ò�Ʒ�к��н϶����ʣ��������Ρ��衢�ơ�������ʣ����һ�����ƣ��������������Ʒ��Ҫ��[5-6]��Ŀǰ�������ϣ��˴���ת���ķ�����Ҫ��ʪ���ɷ�����[7]��ʪ�����յ���Ҫ������ͨ���ܽ⡢��ȡ�����ӽ����ȷ�ʽ��ȥ�������е����ʣ�����ô�Cameco������Areva�ȹ�˾�����ô˷����ɷ������������������ϻ�ѧ��˾������һ���Ʊ�������ֱ�Ӷ�ԭ�Ͻ��л�ԭ����������ת�������Σ�ͨ��UF6�ľ���ȥ������[8]����ȸɷ����գ�ʪ�����ն�����Ҫ��ϵͣ�����ͨ�Ը���ʹ�÷�Χ���㣬��Ŀǰ�������˴���ת������������·�ߣ����㷺Ӧ�����˵��ᴿ[9-11]���߷ŷ�Һ����[12-13]�ȡ�
������泥�ADU����U3O8���˿�Ũ�����������Ҫ��ʽ[14]��ADU��U3O8�������ܽ�Һ���к�ǿ�������ԣ�����Ҫ��������ADU�����ܽ�Һ�к�������泥���U3O8�����ܽ�Һ�в��������ʡ�Ȼ����Ŀǰ�����к��������ߵ���ȡ�Ա��о�����ˣ��������ADU��U3O8���������ܽ�Һ����ͬ��ȡ�����µ���ȡ���ܽ��жԱȣ�̽��ADU�ܽ�Һ��U3O8�ܽ�Һ��ȡ�������ܵ�����Ϊ�乤ҵ��ȡ������������ͬһ�����߹������ṩһ��������֧�֡�
1 ʵ��
1.1 ��Ҫ�Լ�������
ADU��U3O8��TBP��������������Լ�һ��������ú�ͣ��������ͳ������ᣬ68%���������ѻ�ѧ�Լ�����˾����������
JJ-1�ͽ���������ݹ�����������˾��CIT-3600ʯī����Ԥ����X����ӫ���ǣ���������˵�������˾��ICAP-6500ȫ��ֻ��������������ǣ������ȵ繫˾��
1.2 ����������Һ����ȡ�����Ʊ�
����������Һ�Ʊ������Ƚ�һ�����Ĺ�̬ADU��U3O8�ֱ���Ũ��������ܽ⣬���Ƴɸ�Ũ�ȵ�����������Һ��Ȼ������Ӧ��ȵ�ϡ������Һ������ƿ�н��ж��ݣ����Ƴɶ�Ӧ��Ⱥ���Ũ�ȵ�����������Һ��
��ȡ���Ʊ�����TBP�����ú�Ͱ���30%���������������ȣ�����TBPռ30%��
1.3 ʵ�鷽��
���յ�����ȡȡ�ķ�������������������ȡ�����巽�����£�������ȡһ�����İ�Ҫ�����Ƶ�����������Һ��30%TBP-����ú�����μ����ձ��У�Ȼ�����3 min�����Һ�嵹���Һ©���о��÷ֲ㣬�����ϲ�Ϊ��ȡ��ĺ����л��࣬�²�Ϊ��ȡ�����������ˮ�ࡣ���²�ˮ�����ȡ���������ⶨ��Ũ�ȣ�������ʽ����ADU��U3O8�����ܽ�Һ��������E��
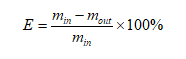 (1)
(1)
ʽ�У�minΪ��ȡǰ��ԭҺ������������������g��moutΪ��ȡ������Һ������������������g��
��ȡҺ���˱��Ͷ�S�ļ��㹫ʽ���£�
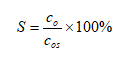 (2)
(2)
ʽ�У�coΪ��ȡ����ȡҺ������������Ũ�ȣ�g/L��cosΪ��ȡҺ��������������ܽ�������ֵΪ130 g/L[15]��
1.4 ��Һ����Ũ�ȷ�������
����g��������������Һ�������ظ���ر���Һ��ͨ��������ԭ��ԭ�����ⶨ��Һ�����������ĺ���������mg��������������Һ��ͨ���������ǽ��в�����
2 ���������
2.1 ��Ũ�ȶ������ʵ�Ӱ��
��������������ȡ��ϵ�У���Ũ�ȶ����յ���������һ����Ӱ�졣������24 �桢ADU��U3O8�����ܽ�Һ���Ϊ1.65 mol/L����ԭҺ����ȡ�����Ϊ1:4.5�������£����첻ͬ����������Ũ����ADU��U3O8�����ܽ�Һ�������ʣ������ͼ1��ʾ��

ͼ1 �ܽ�Һ����Ũ�ȶ������ʵ�Ӱ��
Fig.1 Effect of uranium concentration on yield of uranium
��ͼ1��֪�������ܽ�Һ����Ũ�ȵ�����ADU��U3O8�����ܽ�Һ�������ʾ��������Ӻ����½������ơ�������������л������˺����ϸ�ʱ����ȡ������ϵ������������ڱ��ͣ��������˵Ľ�һ����ȡ����������������½����ӹ�ҵӦ�õĽǶȳ���������˵�Ũ�ȣ��������Ӳ��ܣ�ͬʱ���ٺ��˷�ˮ��ʵ�ֽ��ܼ��ţ�����ܽ�Һ�и�Ũ������ʹ�����������½������½����Ƚ�С����Ũ��Ϊ430 g/Lʱ��ADU��U3O8�����ܽ�Һ���������Էֱ��95.43%��90.34%�������Զ��ԣ�����ܽ�Һ����Ũ���������ġ�
ͬʱ��ͼ1���ɿ���������ͬ�����£�ADU�����ܽ�Һ�������ʽ�U3O8�����ܽ�Һ�ĸߣ���Ҫԭ����ADU�к���笠����ӣ����ܽ�����������ᷴӦ����NH4NO3��������ԭҺ�У�������ȡ��������������������[16]�����ʹ��ADU�ܽ�Һ�������ʽϸߡ�
2.2 ��ȶ������ʵ�Ӱ��
�����Ӱ������������Һ��ȡ�����ʵ�һ����Ҫ���ء���ADU��U3O8�����ܽ�Һ����Ũ��Ϊ430 g/L����ȡ������ԭҺ���Ϊ4.5:1�����£��о���5�ֲ�ͬ�����ADU��U3O8�����ܽ�Һ�������ʣ������ͼ2��ʾ��

ͼ2 �ܽ�Һ��ȶ������ʵ�Ӱ��
Fig.2 Effect of acidity on yield of uranium
��ͼ2��֪��������ȵ����ӣ������ܽ�Һ�������ʾ���֮��ߣ�����ADU�����ܽ�Һ������ٶȽϻ�����ʵ����ȷ�Χ�ڣ�����������93.06%������95.96%����U3O8�����ܽ�Һ�����������ٸ�Ϊ���ԣ�����͵�78.42%�����92.11%����˵����ȶ�U3O8�ܽ�Һ����ȡЧ��Ӱ��ȶ�ADU�����ܽ�Һ��Ϊ������������������ȡЧ�ʣ���������ȵ���1.0 mol/Lʱ��������������ʵ�Ч�������������������Ϊ��Ƚϵ�ʱ�����������������ڴٽ�U3O8�����ܽ�Һ��ȡƽ������ƣ���������߽Ͽ졣��ˣ���U3O8�ܽ�Һ����ȡ��������Ȳ�ӦС��1.0 mol/L��
��ȶ�����U3O8�����ܽ�Һ�������ʽ�ADU�����ܽ�Һ�ߵ�ԭ������������ȡ����������������������ƣ�Ҳ�����������á�����U3O8�����ܽ�Һ��û��NH4NO3�������������Ĵ��ڣ������������Ϊ����������ʱ��U3O8�����ܽ�ҺӰ�����������Ҳͬʱ������ADU�����ܽ�Һ��U3O8�����ܽ�Һ�������ʸߵ�ԭ����ΪADU�����ܽ�Һ�������е���������������������ã�����������ٽ�����ȡ�����е�����ЧӦ�����ADU�����ܽ�Һ��U3O8�����ܽ�Һ�������ʸߡ�
2.3 ��ȶ������ʵ�Ӱ��
��ADU��U3O8�����ܽ�Һ��Ũ��Ϊ430 g/L�����Ϊ1.65 mol/L�����£��о���ȡ������ԭҺ����ȶ������ʵ�Ӱ�죬������ڱ�1���ɱ�1��֪���ܽ�Һ������������ȵ����Ӷ����ӣ����Ϊ4.0ʱ��ADU�����ܽ�Һ�������ʴ�93.21%���������ʵ�����������ȵ����ӣ������������仺����˵�������ʲ���������ȵ�������������ӵġ��ӱ�1Ҳ�ɿ���������ͬ������£�ADU�����ܽ�Һ����ȡЧ������U3O8�����ܽ�Һ��
��1 ��ȶ������ʵ�Ӱ��
Table 1 Effect of flow ratio on yield of uranium
��� | ������ | |
ADU�ܽ�Һ | U3O8�ܽ�Һ | |
3.0 | 80.44 | 75.69 |
3.5 | 88.46 | 84.86 |
4.0 | 93.21 | 88.13 |
4.5 | 95.43 | 90.34 |
��ͬ����£������ܽ�Һ����ȡ�����˵ı��Ͷ����ڱ�2���ɱ�2�ɿ��������Խ�ͣ�����Ӧ���˱��Ͷ�Խ�ߣ����Ϊ3.0ʱ���˵ı��Ͷ�Ϊ88.06%�������Ͷȵ���ߣ�ȴ�����������ʵ���ߣ�����֮��ʷ����ϵ��������Ϊ��ȡҺ���˺��������ӣ��谭����ȡƽ��Ľ�һ�����ƣ��Ӷ������������ʡ���Ҳ������������ȵ����ӣ���ȡƽ����һ�����ƣ�������������ӣ�����ȡ���ջ�ﵽƽ�⣬��������ʵ������仺����������һ������Ķ�ֵ��
����ڽ��ж༶������ȡ�Ĺ����У�Ϊ������ȡЧ�ʣ���Ȳ�Ӧ̫С������ᵼ���˱��Ͷȹ��ߣ��Ӷ����������ʣ������Ҳ��Ӧ���ߣ�����ᵼ����ȡ���������ʽ��͡�
��2 ��ͬ�������ȡҺ�ļ��˱��Ͷ�
Table 2 Uranium concentration and saturation of extraction in different flow ratios
��� | ���Ͷ�/% | |
ADU�����ܽ�Һ | U3O8�����ܽ�Һ | |
3.0 | 88.06 | 83.42 |
3.5 | 83.55 | 80.16 |
4.0 | 77.05 | 72.85 |
4.5 | 70.11 | 66.37 |
2.4 ��ȡ�����������ʵ�Ӱ��
Ϊ�о���ȡ�������������������ܽ�Һ�����ʵ�Ӱ�죬ѡ��������ȡ������Ũ��Ϊ420 g/L�����Ϊ1.65 mol/L��ADU��U3O8�����ܽ�Һ�ֱ���ж༶������ȡ����������ÿ����ȡ���ˮ����Ũ�ȣ������ݸ���Ũ�ȼ��������ʣ����ڱ�3��
�ӱ�3�ɿ�����ADU�����ܽ�Һ����ȡЧ�ʸ���U3O8�����ܽ�Һ���������������ȡʱ���������ѳ�����U3O8�����ܽ�Һ��7����ȡ�����ʡ�����ȡ7�κ������ܽ�Һ�������ʶ��ϸߣ��ֱ�Ϊ99.998%��99.990%������ע�������ADU�����ܽ�Һ���е�5����ȡʱ��ˮ����Һ�п�ʼ�������黯����7����ȡʱ��ˮ�����н϶��黯����黯���Ϊ��һ�����п���������Ƚϵ͵�����£�������������Һ�е�笠������γ������������µġ�Ϊʹ��Ũ�ȷ�����Ϊȷ��ÿ����Ũ�Ⱦ�����ʯī����Ԥ����X����ӫ���ǽ��з������÷�����ƫ��Ϊ5%����֮��Աȣ�U3O8�����ܽ�Һ��7����ȡ������δ�����黯����
��3 �༶��ȡ����ԭҺ����Ũ�Ⱥ�������
Table 3 Yield of uranium under multistage extraction for original extraction solution
��ȡ���� | ADU�����ܽ�Һ | U3O8�����ܽ�Һ | ||
��Ũ��/��mg��L-1�� | ������/% | ��Ũ��/��mg��L-1�� | ������/% | |
1 | 22.70��103 | 94.595 | 40.00��103 | 90.476 |
2 | 338.74 | 99.919 | 2.71��103 | 99.355 |
3 | 16.80 | 99.996 | 458.00 | 99.891 |
4 | 10.13 | 99.997 | 152.47 | 99.964 |
5 | 9.67 | 99.997 | 94.26 | 99.777 |
6 | 5.24 | 99.998 | 44.54 | 99.894 |
7 | 4.42 | 99.998 | 43.42 | 99.990 |
2.5 ��ȡ����Һ�����ʾ�����Ӱ��
ѡ����Ũ��Ϊ420 g/L�����Ϊ1.65 mol/L��ADU��U3O8�����ܽ�Һ��Ϊ��ԭҺ������ԭҺ���е�����ȡ��Ȼ�����ȡ����л�����е������ͣ����������������ͻص�ˮ���У�ͨ�������Ƿ���ˮ����Һ�е�����Ԫ�أ�����15�ֺ����ϸߵ�����Ԫ�صķ���������ڱ�4���ɱ�4��֪������Cr��Cu��Fe��Mn��Mo��Na��Ni��Ti��V��W��As��10������Ԫ����ADU�����ܽ�Һ�еĺ�������U3O8�����ܽ�Һ�еģ�����5������Ԫ�غ�������U3O8�����ܽ�Һ�еġ��ӱ�4���ɿ�����ADU����Һ�е�����Ԫ�غ����ձ����U3O8����Һ�еģ�ADU�����ܽ�Һ������ȥ���ʸ���U3O8�����ܽ�Һ�����ң�ADU�����ܽ�Һ�г�Cr��Mn�⣬��������ͨ��������ȡ����ȥ���ʻ�����91%���ϣ���W��Cr��Mn��������Һ�еĺ������ڹ��������IJ������ޣ������ȷ�������ȥ���ʣ�ͨ����ת�������м��Ʒ����������ָ��Ա�[17]��֪������������Ԫ�صĺ�����������Ҫ�������ϣ�ADU�����ܽ�Һ������ȥ��Ч������U3O8�����ܽ�Һ��
��4 ��Һ������Ԫ�صĺ���
Table 4 Content of impurity element in solution
���� Ԫ�� | ADU�����ܽ�Һ | U3O8�����ܽ�Һ | ||||
��ԭҺ ���ʺ��� /(��g��g-1��U��) | ����Һ ���ʺ��� /(��g��g-1��U��) | ����Ч��/% | ��ԭҺ ���ʺ��� /(��g��g-1��U��) | ����Һ ���ʺ��� /(��g��g-1��U��) | ����ȥ����/% | |
Al | 471.5 | 17.2 | 96.35 | 199.5 | 20.9 | 89.52 |
Ca | 880.1 | 8.1 | 99.08 | 205.1 | 82.7 | 59.68 |
Cr | 3.9 | <2.5 | >35.90 | 9.9 | <2.5 | >74.75 |
Cu | 1.0 | <0.5 | >50.00 | 4.5 | 0.6 | >86.67 |
Fe | 1162 | 40.7 | 96.50 | 2289 | 91.3 | 96.01 |
K | 21.3 | 0.8 | 96.24 | 20.2 | 4.7 | 76.73 |
Mg | 94.9 | 3.8 | 96.00 | 68.2 | 8.8 | 87.10 |
Mn | 2.8 | <0.5 | >82.14 | 3.3 | <0.5 | >84.85 |
Mo | 69.6 | <0.5 | >99.28 | 93.5 | 2.6 | 97.22 |
Na | 133.6 | 5.7 | 95.73 | 192.3 | 19.5 | 89.86 |
Ni | 1.0 | <0.5 | >50.00 | 8.6 | <0.5 | >94.19 |
Sr | 24.9 | 0.8 | 96.79 | 43.6 | 1.4 | 96.79 |
Ti | 6.0 | <0.5 | >91.67 | 8.2 | 0.6 | 92.68 |
V | 11.9 | <0.2 | >98.32 | 14.0 | <0.2 | >98.57 |
W | 2.1 | <0.5 | >76.19 | 2.6 | <0.5 | >80.77 |
As | 17.7 | <0.5 | >97.17 | 21.3 | 1.4 | 93.43 |
2.6 ����臨��ܽ�Һ��ȡ�����ʵ�Ӱ��
ADU�����ܽ�Һ��U3O8�����ܽ�Һ��Ҫ��������ADU�������ܽ�Ĺ����лᷴӦ��������泥�Ϊ̽������臨���ȡ��Ӱ�죬����ͬ����£������Ϊ0.28 mol/L��1.00 mol/L��1.68mol/L��3��U3O8�����ܽ�Һ�зֱ���벻ͬŨ�ȵ�����泥�������е�����ȡ�������������ڱ�5��
��5 ��ͬ����狀�����U3O8�����ܽ�Һ��������
Table 5 Yield of uranium of Nitric Solution of U3O8 with different concentrations of NH4NO3
NH4NO3����/(g��L-1) | ������/% | ||
���0.28mol��L-1 | ���1.00mol��L-1 | ���1.68mol��L-1 | |
0 | 78.42 | 89.52 | 90.34 |
36 | 83.76 | 91.35 | 92.51 |
72 | 88.35 | 92.81 | 93.98 |
108 | 90.28 | 93.22 | 95.02 |
144 | 93.06 | 94.94 | 95.43 |
�ӱ�5�ɿ�������3������£�U3O8�����ܽ�Һ��������������狀��������Ӷ�������������Ƚ�Ϊ���ԣ���˵��NH4NO3Ũ�ȵ���144 g/Lʱ������������������ȡ���������������ã����������������������ȡЧ�ʡ�������ͬ����ЧӦ����ģ�����Һ��NH4NO3�������������������������������ķֽ⣬�Ӷ���������������������ʡ�
3 ����
1�� ����ͬ�����£�ADU�����ܽ�Һ�������ʸ���U3O8�����ܽ�Һ����ȡЧ�ʽϸߡ�
2�� ADU��U3O8�����ܽ�Һ������������������Ũ�ȵ����ӳ������Ӻ��С�����ƣ���ʼʱ�����Ͽ죬�ﵽ��ֵ��ʼ�½����������ϻ�����
3�� ADU��U3O8�����ܽ�Һ����������һ����Χ������Ⱥ���ȵ����Ӷ����ӡ�
4�� ADU��U3O8�����ܽ�Һ��7����ȡ������ˮ������Ũ�ȷֱ�Ϊ4.42 mg/L��43.42 mg/L�������ʷֱ��99.998%��99.990%��
5�� ADU�����ܽ�Һ������ȡ���͵�����ȥ���ʸ���U3O8�����ܽ�Һ��������ȥ���ʻ�����91%���ϡ�
6�� ��NH4NO3Ũ�ȵ���144g/L��ʱ��NH4NO3��������������ȡ�����������á�
�ο����ף�
[1] �����,��. WNA���������ȼ���г�δ������չ������[J]. ����:���������,2007(10):22-22.
[2] ���˴�,������,�ܷ�,��. �ܼ���ȡ���ڷ�ȼ�Ϻ����е�Ӧ�����о�����[J]. ԭ���ܿ�ѧ����,2001,35(5):50-55.
Yang Xingchun, Bao Borong, Zhou Fang, et al. The application and research overview of solvent extraction in spent fuel after treatment[J]. Atomic Energy Science and Technology, 2001, 35(5): 50-55(in Chinese).
[3] ������,�Ⱦ���,������. �˵���ȡ�뾫�ƹ���ѧ[M]. ����:ԭ���ܳ�����m1982:246-258.
[4] ţ���壬�Է�᪣����濥����. ��TBP�ܽ���ȡ���Ʊ��˵紿��������[J]. �˻�ѧ����仯ѧ,2011,33(3)��136-147.
Niu Yuqing, Zhao Fengqi, Shu Zujun, et al. Preparation of nuclear power grade UO2 by TBP solvent extraction technology[J]. Journal of Nuclear and Radiochemistry, 2011, 33(3): 136-147(in Chinese).
[5] ������,����,ׯ����,��. �ҹ���Ȼ�˴��������о��ķ�չ����״[J]. �˿�ұ,1998,17(4):231-238.
Deng Zuoqing, Huang Lunguang, Zhuang Haixing, et al. The development and current status of natural uranium purification technology in China[J]. Uranium Mining and Metallurg, 1998, 17(4): 231-238(in Chinese).
[6] ������,��Ӣ. ��Ȼ�˴���������Һ��ѭ�������о�[J]. �˿�ұ,2018,37(2):108-111.
Gong Daokun, Wang Ying. Study on the recycling of extraction raffinate in the natural uranium purification[J]. Uranium Mining and Metallurg, 2018, 37(2): 108-111(in Chinese).
[7] �η���,������. �����ȼ�Ϻ���[M]. ����:ԭ���ܳ�����,2004:65.
[8] Horng J. Semi-Empirical model for liquid-liquid extraction of UO2(NO3)2�CTBP-kerosene system in acid medium[J]. Ind Eng Chem Process Res Dev, 1984, 23(3): 603-609.
[9] ������,�����,���ط�. ��5208��ȡ����������Һ����ȡ��[J]. �˿�ұ,1991,10(3): 31-38.
Yang Yanxin, Jin Qizhen, Zhang Rongfang. The extraction of uranium from nitric acid with 5208 extractants[J]. Uranium Mining and Metallurg, 1991, 10(3): 31-38(in Chinese).
[10] �����,������,����,��. �����������Һ��ϳɼ����ˣ���������ȡ[J]. ԭ���ܿ�ѧ������2014��48(3):390-394.
Li Hongyu, Shen Lihong��Zhang Le, et al. Synthesis of imidazole-type TSIL and extraction for uranium(��)[J]. Atomic Energy Science and Technology, 2014, 48(3): 390-394(in Chinese).
[11] GIRIDHAR P, VENKATESAN K A, SRINIVASAN T G. Extraction of uranium(��) from nitric acid medium by 1.1 M tri-n-butylphosphate in ionic diluent[J]. Journal of Radioanalytical and Nuclear Chemistry, 2005(1):265��1����31-38.
[12] �߽�,Ҷ��,������,��. �߷ŷ�Һ���ȼ������о���չ[J]. ԭ���ܿ�ѧ����,2013,47(6):99-102.
Gao Jie, Ye Gang, Chen Songzhe, et al. Development of partitioning of Sr from high level liquid waste[J]. Atomic Energy Science and Technology, 2013, 47(6): 99-102(in Chinese).
[13] ������,�γ���,�����. ��ȡ���뷨�����߷ŷ�Һ�Ľ�չ[J]. ԭ���ܿ�ѧ����,2000,34(5):111-115.
Jiao Rongzhou, Song Chongli, Zhu Yongrui. The progress in the treatment of high level waste liquid by extration[J]. Atomic Energy Science and Technology, 2000, 34(5): 111-115(in Chinese).
[14] ������. �˽�������ѧ[M]. ����:�й��˹�ҵ�ܹ�˾�ĩ��ij�����ӡˢ��,1993.
[15] ��ʥ��,�η��� ��ȼ�Ϻ�����ѧ[M]. ����:ԭ���ܳ�����,1995.
[16] ����ǿ,�η���,������. ��ȼ�Ϻ���[M]. ����: �й��˹�ҵ�ܹ�˾�ĩ��ij�����ӡˢ��,1994.
[17] ������,κ��,Ҧ����. ��ת������ѧ[M]. ����:�й�ԭ���ܳ�����,2012.
推荐
-

-

QQ空间
-

新浪微博
-

人人网
-

豆瓣




