����BOPPPS+PBL��ѧģʽ�ķ�����ѧ��ѧ�����ʵ��
��꣬�ܳ����ᄃ�����ɺ�
������������ѧ��ѧ�뻷������ѧԺ������ ���� 130022��
ժ Ҫ��Ϊ����ѧ����ѧϰ��Ȥ��DZ�ܣ���ǿѧ���Ĵ���˼ά��ʵ��������������ڡ�BOPPPS+PBL����ѧģʽ�ķ�����ѧ�γ̽�ѧ�ĸ�Է�����ѧ�γ̵����µ�����������Ϊ����������BOPPPS+PBL��ѧģʽ�ľ���ʵʩ���̡������ѧ���������ǿ�ǰ�����С��κ�ȫ���ã�ͻ����ͬ�����ʦ����������������ˡ���ѧ��Ϊ���ġ��Ľ����������ѧ��ѧϰ�������ԺͲ����ԣ���Ч������ѧЧ����
�ؼ��ʣ�������ѧ��BOPPPS��PBL����ѧ��ƣ���ѧʵ��
��ͼ����ţ�G642 ���ױ�ʶ�룺A
������Ŀ������ʡ������ѧ��ʮ���塱�滮2023���һ����⡶�μⴴ���˲�����Ϊ����ķ�����ѧ�γ̡�BOPPPS+PBL����ѧģʽ�ĸ��о�������Ŀ��ţ�GH23672����
����飺��꣨1990-����Ů�����ֳ����ˣ������ڣ���ʿ����Ҫ�о�����Ϊ������ѧ��
Teaching Design and Practice of Analytical Chemistry based on BOPPPS+PBL Teaching Mode
HOU Juan, ZHOU Chen, SUN Jing, LIN Zihan
(Colledge of Chemistry and Environmental Engineering, Changchun University of Science and Technology, Changchun, 130022)
Abstract: In order to stimulate students' learning interest and potential, strengthen students' innovative thinking and practical ability, the teaching reform of analytical chemistry course based on BOPPPS+PBL teaching mode was put forward. Taking gravimetric analysis in chapter 7 of the analytical chemistry course as an example, this paper expounds the concrete implementation process of the teaching mode. The overall teaching activity design is flexible, covering the whole class, highlighting joint participation and teacher-student interaction, which fully reflects the student-centered education concept. The curriculum reform can stimulate students' learning initiative and participation. The teaching effect has been improved effectively.
Key words: analytical chemistry; BOPPPS; PBL; teaching design; teaching practice
���ſƼ��������ƽ���֪ʶ�����Ͱμ��˲�������Ϊ�����Ĺ����ϵ������������2018��9�¾���ʵʩ����Խһ�μ⡱�ƻ�2.0��ָ���ӡ���Χ�����������ʡ����¡��ȷ�����аμⴴ���˲�������ǿ��Χ�Ƽ���ѧ��ѧϰ��Ȥ��DZ����γ̽�ѧ�ĸ��Ϣ�����ķ�չ�����ڽ�����������Ӧ�ã����ظ�У�����������ƽ����ϡ����»��ʽ��ѧģʽ���ڳ������ѧ����������Ϊ�ص�Ļ����Ͽ�չ֪ʶ���ں��������������ڵ���ѧ���Ļ����ԡ������ԺͿγ̲���ȣ�����ѧ������ѧϰ��Ȥ�ʹ���˼ά������ʵ��ʦ��������֪�к�һ[1-3]��
������ѧ�γ����о�������ɡ������ͽṹ�Ȼ�ѧ��Ϣ��������۵�ѧ�ƣ��ǻ�ѧ�����༰�����רҵ��Ҫ��ѧ�ƻ����γ̣���ѧ��ѧϰ�����ʹ���˼ά�������dz���Ҫ�����ú�֧�ű��ϣ�����Ȼ��ѧ�����̼���Ӧ���в��ɻ�ȱ�Ļ������ߡ����ճ��Ľ�ѧ��֯�����У�������֪ʶ����ϵ����ϵͳ�Ժ�������ΪĿ�꣬����ѧ���ķ�չ��������������ʱ����Ҫ���¿γ���ϵ���ӡ����ݳ¾ɡ���ǰ�ؼ�����ʵ�������ѽڡ���ӳ�ھ����ѧʵ����������Ϊ�γ���ϵ�����ȵ͡���ѧģʽ���ѧ��������ѧ��������ȱ������˼ά�Ϳ�����Ȥ��רҵӦ���Բ�ǿ�����⣬�����ڰμⴴ�����˲ŵ���������ˣ��Լ�ǿѧ������˼ά��ʵ������ΪĿ�꣬������������У���Ƚ���ѧ�����ѧ�Ŷӽ���BOPPPS���͡�PBL����ѧģʽ���ϣ���������-���»��ʽ�ǻ۽�ѧ������Ч����ѧ���Ŀ�ѧ˼ά��רҵ�����Լ�����ʵ������������߽�ѧ�������дٽ����á�
1 BOPPPS+PBL��ѧģʽ
��������ȫ������ߵ�ѧУ̽��������������Ϳ���Ϊ���ĵĽ�ѧģʽ���������Ա�Ϊ�����ĸ��ع顱�Ľ�ѧ�ĸ���ڳ�ֵ���ѧ���Ļ����Ժ������ԣ������䴴��˼ά�����շ������ʵ���������������������Ҳ���ѷ��֣�����2012�꣬���ڽ��������߾����ֽ������ϡ����»��ʽ��ѧģʽ�ĸ��̽���������BOPPPSģ��Դ���ڼ��ô��ʦ������ѵ��������ǿ����ѧ��Ϊ���ģ���Ҫ�����γ̵��루Bridge-in����ѧϰĿ�꣨Learning Objective��������ǰ�⣨Pre-assessment��������ʽѧϰ��Participatory Learning�������ú�⣨Post Assessment�����ܽᣨSummary�������Σ������½�������ͽ��ʷ�Ϊ�������ݵĽ�ѧģʽ��ĿǰBOPPPS��ѧģʽ��ȫ���������ѧУ��ʦ������ѵ�е��Թ㷺Ӧ�ã���һ�ָ�Ч�Ľ�ѧģʽ[4-6]����ģ�Ͳ��ݽ����γ������ıպ���������ѭѧ��Ϊ���ģ�Ŀ��Ϊ����ע�ؽ�ѧ�����뷴���Ľ����ɲ�����ǿ��PBL��ѧ��������Ϊ�����ѧϰģʽ��ƫ��С�����ѧϰ������ѧϰ����������ѧ����Howards Barrows������������ͨ������ѧ��Ϊ���ġ��������Ż���ͳ��ѧ�еı���ѧϰģʽ������ȼ���ѧ��������ѧϰ��Ȥ�ʹ�����[7, 8]��
���ڵ�ǰ��У��ѧ���������»�Ͻ�ѧ�Ĵ������£�̽���µĿ��ý�ѧ��ʽ��ע��ѧ���Ĵ���˼ά��ʵ���������������Ϻõĵ���ѧ���������ԣ��Ƿdz��б�Ҫ�ġ���ͼ1��ʾ�����ǹ�������ѧ��Ϊ���ġ��ġ�BOPPPS+PBL��������ѧ�γ̽�ѧ�ĸ�ģʽ��������ǿѧϰ��Ȥ������ѧϰ�����������γ̵ġ�����һ�ȡ������û�����������֪ʶ��ѧ�Ļ����ϣ�����ѧ���ص��֪ʶ��ϵ�����ԣ��ḻ��ѧ����Խ�רҵ�����ƽ�֪ʶ��ء��ĸ���Ҫ��Ŀ���ƶ�������ѡ��ѧģʽ�Լ���ѧ���۵ȷ�����н������Խ�ʦ�Ŷӽ���ͽ�ѧ��Դ�Ż���Ϊ��֧�ֱ��ϡ����磺��֪ʶ�����������ʡ�˼���ȶ�����������ƶ���ѧĿ�꣬���������ݺ�ѧ�����н����ۺϿ�������ѭѧ����֪���ɺ�רҵ���������н�ѧ��Դ����Ч���ϣ������������רҵӦ�á�ǰ�ؼ�����ѧ�ƽ��棬��ʵ�ַ�����ѧ�γ̵ĺ����������չ���������õĸ߽��Ժ���ս�ȣ��ḻ���û�����ѧ���������Ժͻ����ԣ���ѧ������Ŀ��н���ѧϰ���γɶ�Ԫ������ϵ������ʦ�����������������Ȼ��ƣ��ٽ�ͬѧ֮�䡢С��֮�����Ч�����ල���Բ���ʽѧϰ��������ƽ���ѧ������أ�ʹѧ���ܹ���ȷѧϰĿ�꣬����ѧ����רҵ���š���ѧ������ѧ�����õ�������
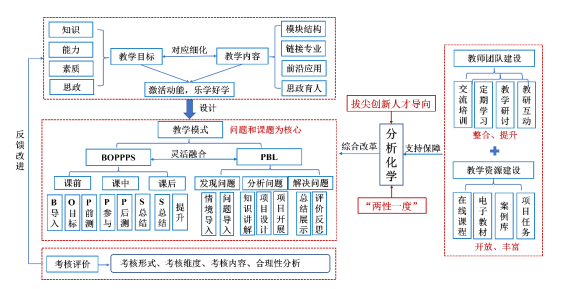
ͼ1 ������ѧ�γ��ۺϸĸ�
Fig. 1 Comprehensive reform of analytical chemistry curriculum
2 BOPPPS+PBL��ѧģʽ�ڷ�����ѧ�γ̽�ѧ�е�ʵ��
����УӦ�û�ѧ�ͻ���רҵ�ķ�����ѧ�γ�Ϊ�������ѧ��Ϊ����ԭ����ѧ�ý�ϣ����л��ڡ�BOPPPS+PBL����ѧģʽ�Ŀ��ý�ѧʵ����������ѧ�γ̵�����Ŀ��Ϊѧϰ������ѧ��������֪ʶ�����ն�������������ԭ�����ص㡢����������Ӧ����������Ϥ�������ݡ�ͳ�ƺͿ��Ƶ�֪ʶ�������ϸ�ġ������ĸ�����ն���������һ�㲽�裬�ܹ����÷�����ѧ����ԭ�������ݴ������������������������ɡ������Ȼ�ѧ��Ϣ��صĸ��ӹ������⣬�����ʽ��ж��Զ��������������Ч���ۣ��ô�����˼ά�жϺͽ��ʵ�����⣬����ʵ�����ǵĿ�ѧ̬�Ⱥ��Ͻ�ϸ�µĹ������硣�γ�ѡ�õĽ̲�Ϊ����������ѧ���Ĵ���ѧ�ϱ�ġ�������ѧ�����߰棬���е�����Ϊ�����������ͳ����ζ����������������Ǵ�ͳ�Ļ�ѧ����������֪ʶ��չ��ǿ�����ݷḻ���������С���BOPPPS��ѧģ����Ϊ��ܣ����Խ����÷�Ϊ�����ν��пγ���ƣ������ڿι���������PBL��ѧ����������Ϳ���Ϊ��������ѧ����������ѧϰ���γ�̽���Ϳ��á�
2.1 �γ̵���Bridge-in
�γ̵���������Ͻ�ѧģʽ����ǰһ���ڳ���ѧϰͨƽ̨������ѧ�����ѧϰ���ݣ�ͨ������ȵ��¼�������ʱ���Լ����а�����������Դ������ѧ����ѧϰ��Ȥ�����������������ԭ����������ʵ��Ӧ�������˼�����������뱾�½�ѧϰ���ݡ�����ķ�ʽ������Ƶ���ϡ�����롢���ʵ����ʵ������ȡ����磺�Ի����������������PM10��PM2.5�IJⶨ���������������������Ļ���ԭ���Ͳ������̣��Բ�ͬ���ͳ�����Ӧ��ͼƬ����Ƶ����ѧ���Գ������ͺͳ����γɹ��̵�˼�����Ծ�����˷��ֲ���ȡ�ء�������ˮ�������Ӻ����ⶨʵ��Ȱ�������������ȵ�Ӱ�������Լ���߳������ȵĴ�ʩ��
2.2 ѧϰĿ��Learning Objective
��������ѧ�ҡ�����ѧ�Ҳ�³ķ˵��������Ч��ѧʼ��֪��ϣ���ﵽ��Ŀ����ʲô�����Ŀ�겻����ʦҪ֪����ѧ��ҲҪ֪���������½�ѧϰĿ��ͨ��ppt�����ֲ����ķ�ʽֱ��չ�ָ�ѧ������ѧ���������ձ��ڿε���Ҫѧϰ�����ѧϰ���ݣ�����Ŀ��н���ѧϰ��������������ѧЧ�ʡ������ֵ�֪ʶĿ��������������������Ļ���ԭ���Ͳ�������ȷ�������������ص㣬�˽����������Գ�����Ҫ������Ӱ������ܽ�Ⱥʹ��ȵ����أ����ճ������γɹ��̣����վ��ͳ�����Ǿ��ͳ������γ�������ͬʱ��Ͽγ�Ŀ��ͱ�ҵҪ��ϣ��ѧ���ܹ��Է�����ѧ�ӽǷ������⣬������������ѧ֪ʶ��������ʹ����ԵĽ��ʵ�����⣬����ǿ������˼ά��������
2.3 ����ǰ��Pre-assessment
ͨ�����ϲ������ѧ�����п���ǰ�⣬�˽�ѧ���Ļ�����Ԥϰ������ܹ�������ʦ�������н�ѧ��ƣ�������ѧ���ȡ�һ��ǰ����Ŀ�Ի���֪ʶ����Ϊ�������Ͷ�����Ϊѡ���⡢�������ж��⡣���ǰ���ѧ�������߲鿴�𰸡������ͷ��������ƽ̨ͳ��ѧ���Ĵ������������������ʦ��ѧ��ͨ������ǰ����Խ��и��Ի���ϣ����Լ�δ���յ����ݽ��н�һ���ĸ�ϰ���̣���ʦͨ������ǰ���˽�ѧ����ѧϰ��������ѵ㣬�ں����Ŀ��ý�ѧ�п���������ԵĿ�չ��ѧ���
2.4 ����ʽѧϰParticipatory Learning
����ʽѧϰ��������ѧ�������Ҫ��һ������Ҫ�������½�ѧ��ʽ���С���ʦ����Ƶ��ppt������������ȶ������Ľ�ѧ��ʽ�����ѵ�������н��⣬���ݿ���ǰ���Լ�ѧ���Ļ������죬����ѧ����չ���Ի�ѧϰ���ص�֪ʶ��ǿ����ϰ��ͬʱ��ϸ��˱��桢С�����ۡ������������龳��ѧ�ȷḻ�Ļ��ʽ����ѧ���������뵽���û�У��Ժ��������ķ�ʽ����֪ʶ����������գ�������չ֪ʶ�ĺ��������죬�γɴ�����˼ά�����磬�ڽ���Ӱ��������ȵ����ص��½�����ʱ�����Ը��������������������ѧ�������úϺ���������Ҫ��ij��������壬ͨ��С�����۹������ͺ�����Է��������Ӱ�죬�Աȷ�������������ԭ������λ����Ӧ�Ĵ�����ʽ��ͨ��С��㱨������ѧϰ�����������ܽ�����ٳ���մ�۵ķ�����
2.5 ���ú��Post Assessment
���ݽ�ѧĿ��ͽ�ѧ���ݣ���ѧϰͨ����ƽ̨����������Ŀ������ѧ����ڿε����ճ̶Ⱥ��������������������Ҫ��ʽ����ѡ���⡢����⡢����⡢ʵ����Ƽ���Ŀ�⡣����ѡ���⡢�����ͼ������Ҫ����ѧ���Ի�������֪ʶ�����������ͬʱ�����ֲ�ε�ʵ����ƺ���Ŀ������Ŀ������ҹ��������Ʊ������մɲ��ϵ����ף��ó������۶Ը÷������н��ͺͷ�������ƹ�ҵ�Ȼ��������ⶨ��ʵ�鷽�������Ը÷�������ȱ����м�Ҫ�������Դ˼���ѧ������Ӧ�ó������������ʵ�������������ѧ�����ϴ��Ⲣ������ʱ���Ĵ𰸺ͽ������������¸�ϰ��ƽ̨�����������������ʦ�����½ڿ��Ͻ���С��㱨������ԵĽ��⡣
2.6 �ܽ�Summary
�κ��ܽ�������ѧ��������䣬����γ����ݵ�֪ʶ���������ϵ����������ѧ���������õ�ѧϰϰ�߾�����Ҫ�����á��γ̽��ںͲ��Խ�����ʦ��������ѧ������֪ʶ����ͼ��˼ά��ͼ����һ��ǿ�������������ڽ��ʵ������ʱ��ע������Ͳ������̡���ʦͨ�������������ҵ����������������Ŀ�ͷ�������������ѧ����ʵ�ʷ���������з�����ƺ���������ѧ���γɽ���ʽ˼ά�����⣬���ڿ��ű���ʵ���Һ��о���ʵ���ң�ѧ������������Ŀ��Ʒ�������ʵ������ͷ�����ͬʱ����Ҳ����ѧ�����������ѧ������ѵ����Ŀ���߷�����ѧ������ľ�����й���������֪�к�һ��
2.7 PBLʽѧϰ
�ڽ�ѧ��У��ᴩ������龳������ѧ��������չѧϰ����������֪ʶ��ʵ�����γ�̽���Ϳ��á�����������У���ʦ��ѧ����ͬѰ������Ľ������������ѧ���ڻ�IJ����������ʵ�֪ʶ�����ת�����������˵��ۺ����������磺�ھ�������������У���ϵ�½�������ѧ��������⣬�����������������Щ�������������γɵij�������Щ�ص㣿���Խ���Ӧ������Щ���й����У����ײ��ϵĺϳɹ������õ�����Щ����֪ʶ���������Ƴ��������Ӷ��õ���Ҫ�ij������ͣ�ѧ�����ݽ�ʦ�����⣬�ڿ���ʱ��ͨ�����ײ��ĺ�С�����ۣ�Ѱ�Ҵ𰸣��γ�����Ľ���������ڿ����ϻ㱨����չ���ں���以������PBLģʽ�£�ѧ���ӱ���ѧϰת��Ϊ����̽�����ܹ��ھ���˽�ѧ��ǰ��֪ʶ����ǿ����ѧϰ��ʶ��������۵�ʵ������������
3. ����
BOPPPS+PBL��ѧģʽ��עѧ��̽�����������ʹ���˼ά���������Թ�ͬ�����ʦ������Ϊ�������ٽ�ѧ����չ���ѧϰ����ǿ��֪ʶ�����ͨ���κ�����ѧ���ʾ����飬ѧ���Ըý�ѧģʽ���۽ϸߣ�������֤�˸ý�ѧģʽ�Ŀ����ԡ��ڷ�����ѧ�γ̺����ĸĸ�����У����ǽ���һ���ḻ�γ̰������о���Ŀ��������ʦ�����Ŀ��кͽ�ѧˮƽ�������������õķ������ƣ��Ա�Է��ֵ�����������ʱ������ȡ�ø��õĽ�ѧЧ����
�� �� �� ��
[1] ���Ľ�, ����, ̷ѧ��, ��. �������������Ĵ�ѧ��ѧ����Ρ����ʽ��ѧģʽ̽��[J]. �߽�ѧ��, 2023, 9(27): 130-133.
[2] ��ά, ����, ����. Ӧ���ͱ���ԺУ����ѧ���ʽ��ѧ̽����ʵ��[J]. ��������, 2023, (25): 40-43.
[3] ����, ����, ����, ��. ������ѧ�γ��������»��ʽ��ѧ[J]. �й�ұ�����, 2022, (06):33-35.
[4] ������, ����. ���ڡ�ѧϰͨ+BOPPPS��������������ѧ�γ����Ͻ�ѧ���[J]. �������ʽ���, 2023, 9(17):157-160.
[5] �J÷, ����, ����, ��. ��������ú�BOPPPS �Ľ�ģ�͵Ļ��ʽ��ѧ�����ʵ����������ϸ�ϳɵ�Ԫ��Ӧ[J]. ��ѧ����(��Ӣ��), 2023, 44(18):37-44.
[6] ��ͤͤ, �߽���, ������. �������»��ʽBOPPPS��ѧģʽ���л���ѧ���ý�ѧ�е�Ӧ��[J]. ����ʦ����ѧѧ��, 2023, 42(6): 177-180.
[7] ¬��, �¾���, ���, ��. PBL��CBL��ѧģʽ�ڡ����ﻯѧ���γ̽�ѧ�ĸ��е�̽��[J]. �����Ļ�ѧ, 2019, 39(6):1275-1278.
[8] ����, κ����, ����. ����PBL��ҩѧרҵ��������ѧ���γ̽�ѧ̽��[J]. ������ѧ��̳, 2023, (19):129-132.
推荐
-

-

QQ空间
-

新浪微博
-

人人网
-

豆瓣




